Thermal Tolerance of Native Fish Species
The internal temperature of fish, and their physiological development, is regulated by their thermal environment. Species are adapted to a particular thermal regime, establishing timing of development and life stage transitions (Steel et al. 2017). The influence of ambient temperature on the rate of physiological processes leaves fish vulnerable to changes in temperature. All ectotherms have a thermal threshold or upper thermal tolerance limit at which particular biological processes breakdown, leading to reduced physiological performance. This threshold is most likely related to the thermal history to which they have been exposed and are adapted (Comte and Olden 2017, Eliason et al. 2011).
In order to understand the physiological capacity of fish relative to temperature, experiments are conducted to estimate metabolic rate, the rate at which an organism assimilates and expends energy. We employ whole-animal respirometry (Svendsen et al. 2016), measuring the oxygen consumption (MO2) of individual fish in an enclosed chamber. Using this method, we calculate standard metabolic rate (SMR), maximum metabolic rate (MMR), and most relevant, aerobic scope (AS). Aerobic scope (AS) is the energy required by an organism to undertake activities beyond those required just simply to exist. Uniquely, this research is being conducted using an 8-chamber intermittent-flow respirometry system set up adjacent to the stream where fish are collected. This approach allows us to minimize stress to the fish by requiring little to no transportation and reduces acclimation and holding time as compared to the similar studies conducted in a laboratory setting.
In the 8-chamber intermittent-flow respirometry system (Figure 1), each chamber is equipped with 2 water pumps. One pump flushes fresh water through the chamber and the other pump recirculates water continuously past an oxygen electrode that monitors oxygen levels in the chambers. All the chambers and pumps are submerged in large fiberglass tanks. The tanks will receive either ambient water from the surrounding waterbody via a water pump or heated water from an adjacent header tank that is equipped with a water heater. This system allows eight fish to be tested simultaneously.
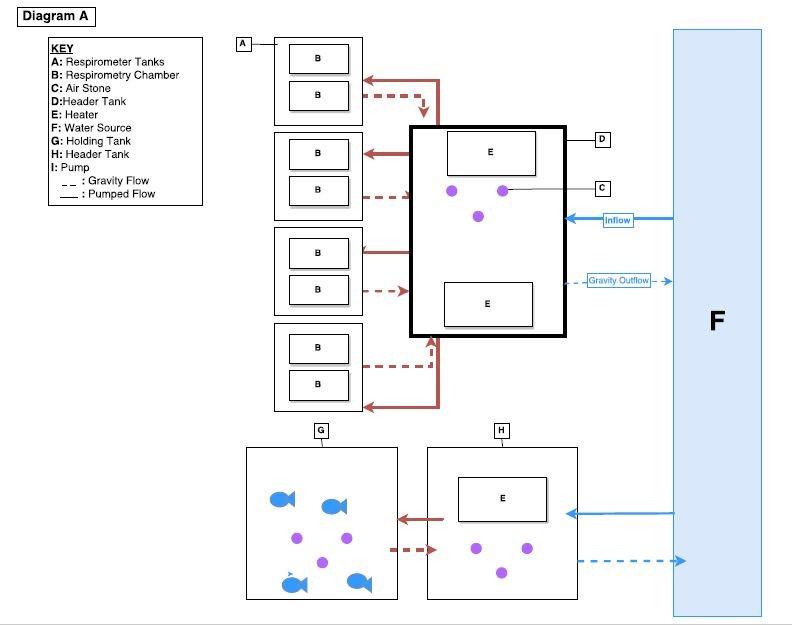
Figure 1. Diagram of 8-chamber intermittent-flow respirometry system.
This project is being conducted in collaboration with Dr. Erika Eliason at University of California, Santa Barbara.
Comte, L. and J.D. Olden. 2017. Evolutionary and environmental determinates of freshwater fish thermal tolerance and plasticity. Global Change Biology 23, 728-736.
Eliason, E.J., T.D. Clark, M.J. Hague, L.M. Hanson, Z.S. Gallagher, K.M. Jeffries, M.K. Gale, D.A. Patterson, S.G. Hinch, and A.P. Farrell. 2011. Differences in thermal tolerance among sockeye salmon populations. Science, 332 (6025), 109-112.
Steel, E.A., T.J. Beechie, C.E. Torgersen, A.H. Fullerton. 2017. Envisioning, quantifying, and managing thermal regimes on river networks. BioScience 67: 506-522.
Svendsen, M., P. Bushnell, J. Steffensen. 2016. Design and setup of intermittent-flow respirometry system for aquatic organisms. J Fish Biol 88:26–50.